General scientific interest:
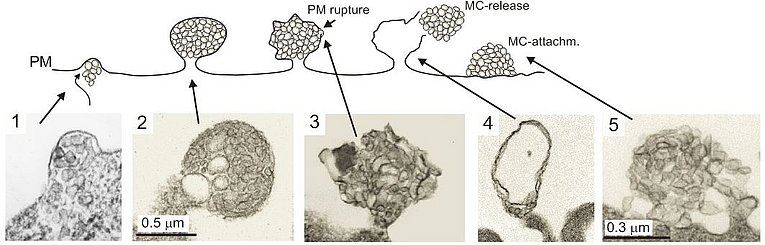
Our scientific interest centers on the role of plasma extracellular vesicles (pEV) in the pathogenesis of human diseases, specifically HIV infection, malignant Melanoma and Alzheimer's disease. Studying the pathogenesis of HIV infection, in particular the role of the viral Nef protein, we discovered that Nef expression leads to the secretion of vesicles clusters originating from the endosomal compartment (Muratori et al., 2009). Importantly the vesicles could be found in high concentrations in the bloodstream of infected individuals (Lee et al., 2016). We then discovered that increased concentrations of pEV are also associated with other diseases including Melanoma (submitted). From these findings we deduced that pEV play an important role in the pathogenesis of many human diseases and hence we concentrate our scientific work on the molecular regulation of pEV, their content and function in target cells.
HIV infection:
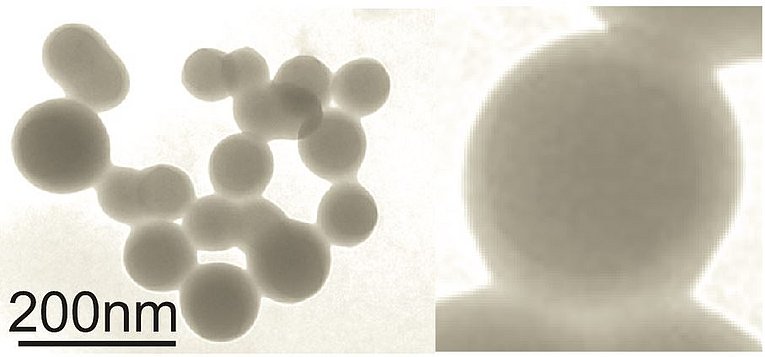
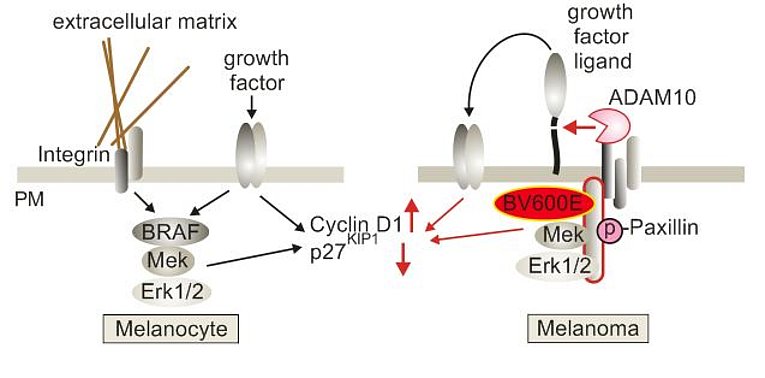
Our lab was the first to describe that HIV infection, specifically expression of the Nef protein, leads to the massive secretion of vesicles clusters of endosomal origin (Muratori et al., 2009). In a subsequent study we analyzed and described the molecular mechanism that leads to the secretion of these vesicles (Lee et al., 2013). We found that integrins and the adapter protein Paxillin play a pivotal role in this context. In this paper we also describe that active ADAM17 and 10 proteases are uploaded along with HIV Nef. In a recently published paper we demonstrate that these vesicles persist in high concentrations in the plasma of infected individuals despite effective antiviral therapy (Lee et al., 2016). Importantly the concentration of Nef and ADAM17 in pEV correlated stringently with the CD4 helper T cell level. In a recently submitted paper we describe how these vesicles causes a massive secretion of TNF from endosomal compartments (Wittki et al., in revision). This secretion mechanism was highly unusual as TNF is believed to be shed from the plasma membrane. TNF is arguably the most important factor stimulating HIV replication and has an important role in the development of many HIV comorbidities. In an upcoming paper we describe how and where these pEV are secreted in infected individuals.
Group Leader, MD, PhD, Prof and former Associate Professor at the Depatment of Immunology and Microbiology at the Miller School of Medicine in Miami, currently attending Physician and Scientist at the Department of Dermatology in Erlangen. Medical Studies in Erlangen, Postdoc in San Francisco, UCSF.
Leitung
Apl. Prof. Dr. med. Andreas Baur MD, PhD
Research Group Leader
Telefon: 09131 85-39537 09131 85-39534
E-Mail: andreas.baur(at)uk-erlangen.de
Malignant Melanoma:
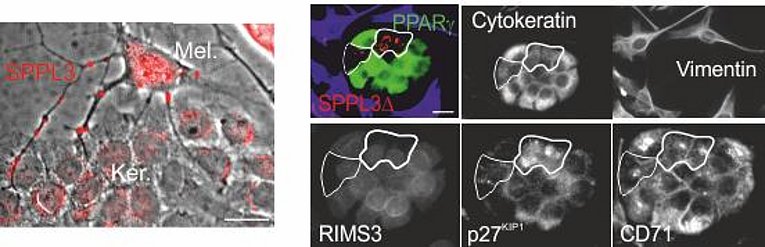
In the course of our HIV projects we also analyzed plasma samples from melanoma patients. We discovered that also these patients have elevated levels of pEV containing ADAM10 protease. Surprisingly residual levels of these pEV can be detected many years after the primary tumor has been removed. In the last years we have investigated these findings in great detail and came to some surprising conclusions. It seems that the innate immune system is sensing the presence of tumor cells and mounts a pEV-mediated response to suppress tumor growth and the formation of metastases. Currently we are investigating the molecular details of this regulation. The first part of our results, which have been established in close collaboration with the group of Julio Vera-Gonzales, has been submitted for publication (Dindorf et. al., submitted).
Alzheimer's disease:
In close, collaboration with researchers and clinicians from the Department of Psychiatry in Erlangen we analyzed pEV from Alzheimer patients. Initial findings were quite exciting and we are currently investigating the role of these vesicles in more detail. We expect our first publication on this subject to be submitted at the end of the year.
Extracellular vesicle biomarkers:
Extracellular vesicles in plasma contain large number of biomarkers including micro-RNA, proteins, cytokines/chemokine/soluble factors (CCF) and enzymes, in particular proteases. We discovered that in many human diseases pEV are shed into the periphery containing a characteristic barcode like pattern of these markers. These pEV barcodes could be used to screen and monitor human diseases. Indeed, we have completed first clinical studies that confirm this assumption. Hence pEV-derived biomarkers have a promising future in translational medicine. Hence one of our research goals is to understand, which of these markers are secreted and for what reason. We have already published how these barcodes look like (Lee et al., 2016) and are currently preparing additional publications. Based on this work we filed several patents and start to collaborate with biotech companies to exploit this highly promising lead.
Reference List
Baur,A.S., Lutz,M.B., Schierer,S., Beltrame,L., Theiner,G., Zinser,E., Ostalecki,C., Heidkamp,G., Haendle,I., Erdmann,M., Wiesinger,M., Leisgang,W., Gross,S., Pommer,A.J., Kampgen,E., Dudziak,D., Steinkasserer,A., Cavalieri,D., Schuler-Thurner,B., and Schuler,G. (2013). Denileukin diftitox (ONTAK) induces a tolerogenic phenotype in dendritic cells and stimulates survival of resting Treg. Blood 122, 2185-2194.
Dindorf,J., Lee,J.H., Eberhard,M., Xin,L., Ostalecki,C., Koliha,N., Blume,K., Wild,S., Schuler,G., Vera-Gonzales,J., and Baur,A.S. (2016). Cancer cells induce a systemic extracellular vesicle (EV)-mediated suppressive immune response (sumitted).
Lee,J.H., Schierer,S., Blume,K., Dindorf,J., Wittki,S., Xiang,W., Ostalecki,C., Koliha,N., Wild,S., Schuler,G., Fackler,O.T., Saksela,K., Harrer,T., and Baur,A.S. (2016). HIV-Nef and ADAM17-Containing Plasma Extracellular Vesicles Induce and Correlate with Immune Pathogenesis in Chronic HIV Infection. EBioMedicine. 6, 103-113.
Lee,J.H., Wittki,S., Brau,T., Dreyer,F.S., Kratzel,K., Dindorf,J., Johnston,I.C., Gross,S., Kremmer,E., Zeidler,R., Schlotzer-Schrehardt,U., Lichtenheld,M., Saksela,K., Harrer,T., Schuler,G., Federico,M., and Baur,A.S. (2013). HIV Nef, paxillin, and Pak1/2 regulate activation and secretion of TACE/ADAM10 proteases. Mol. Cell 49, 668-679.
Muratori,C., Cavallin,L.E., Kratzel,K., Tinari,A., De,M.A., Fais,S., D'Aloja,P., Federico,M., Vullo,V., Fomina,A., Mesri,E.A., Superti,F., and Baur,A.S. (2009). Massive secretion by T cells is caused by HIV Nef in infected cells and by Nef transfer to bystander cells. Cell Host. Microbe 6, 218-230.
Ostalecki,C., Lee,J.H., Wittki,S., Dindorf,J., Collenburg L., Schierer,S., Simon,B., Schliep,S., Kremmer,E., Schuler,G., and Baur,A.S. (2016). Multi-Epitope Tissue analysis reveals SPPL3-mediated ADAM10 Activation as a Key Step in the Transformation of Melanocytes (submitted).
Wittki,S., Ostalecki, Lee,J.H., C., Geist, MM., Tibroni, N., Harrer,T., Schuler,G., Fackler,O.T., and Baur,A.S.
HIV Nef- and Notch1-dependent endocytosis of ADAM17 induces vesicular TNF secretion in chronic HIV infection (EBioMedicine, in revision).
Other significant publications
Baur,A.S. (2011). HIV-Nef and AIDS pathogenesis: are we barking up the wrong tree? Trends Microbiol. 19, 435-440.
Baur,A.S., Sass,G., Laffert,B., Willbold,D., Cheng-Mayer,C., and Peterlin,B.M. (1997). The N-terminus of Nef from HIV-1/SIV associates with a protein complex containing Lck and a serine kinase. Immunity. 6, 283-291.
Baur,A.S., Sawai,E.T., Dazin,P., Fantl,W.J., Cheng-Mayer,C., and Peterlin,B.M. (1994). HIV-1 Nef leads to inhibition or activation of T cells depending on its intracellular localization. Immunity. 1, 373-384.
Fackler,O.T. and Baur,A.S. (2002). Live and let die: Nef functions beyond HIV replication. Immunity. 16, 493-497.
Witte,V., Laffert,B., Rosorius,O., Lischka,P., Blume,K., Galler,G., Stilper,A., Willbold,D., D'Aloja,P., Sixt,M., Kolanus,J., Ott,M., Kolanus,W., Schuler,G., and Baur,A.S. (2004). HIV-1 Nef mimics an integrin receptor signal that recruits the polycomb group protein Eed to the plasma membrane. Mol. Cell 13, 179-190.
Wolf,D., Witte,V., Clark,P., Blume,K., Lichtenheld,M.G., and Baur,A.S. (2008). HIV Nef enhances Tat-mediated viral transcription through a hnRNP-K-nucleated signaling complex. Cell Host. Microbe 4, 398-408.
Wolf,D., Witte,V., Laffert,B., Blume,K., Stromer,E., Trapp,S., D'Aloja,P., Schurmann,A., and Baur,A.S. (2001). HIV-1 Nef associated PAK and PI3-kinases stimulate Akt-independent Bad-phosphorylation to induce anti-apoptotic signals. Nat. Med. 7, 1217-1224.
Xu,X.N., Laffert,B., Screaton,G.R., Kraft,M., Wolf,D., Kolanus,W., Mongkolsapay,J., McMichael,A.J., and Baur,A.S. (1999). Induction of Fas ligand expression by HIV involves the interaction of Nef with the T cell receptor zeta chain. J. Exp. Med. 189, 1489-1496.